Age-Related Wet & Dry Macular Degeneration (AMD)
This content is also available in:
Español (Spanish)

Are you experiencing blurring or a blind spot in the center of your vision? Are you seeing blurry areas on a printed page? Do some straight lines appear wavy? Are there dark spaces or areas in the center of your vision? It might be age-related macular degeneration.
Age-related macular degeneration (AMD) is a gradual, progressive, painless deterioration of the macula, which is the small area in the center of the retina that gives us our detailed vision. This is why someone with vision loss from macular degeneration may have trouble reading mail or newspapers but have no trouble spotting an object off to the side or while walking around, even in unfamiliar places.
This ability to see everything around a room but not to see the very thing one is looking at is confusing to others. This is partly because we consider vision a dichotomy of full sight vs. blindness.
Individuals with age-related macular degeneration are “in between.” They may not have full sight, but they are certainly not blind and never will be from AMD. They have low vision, or an even better description is that they are “hard of seeing,” a term coined by the late Dr. Lorraine Marchi, Founding Director of the former National Association for Visually Handicapped. Like the familiar term “hard of hearing,” it sounds more manageable and accurate.
Symptoms of AMD
Your eye care specialist will likely see signs of macular degeneration before you are aware of any vision loss. When you do start experiencing vision loss from age-related macular degeneration, symptoms can include:
- Blurred or “fuzzy” vision
- Straight lines, such as sentences on a page, appear wavy or distorted
- Blurry areas on a printed page
- Difficulty reading or seeing details in low light levels
- Extra sensitivity to glare
Simulation of Vision Loss from AMD
Here are some simulations of how individuals with vision loss from age-related macular degeneration would see various scenes. They were created by David J. Marmor, MFA and Michael F. Marmor, MD, and published in Archives of Ophthalmology, 2010; 128:117-125. (Used with permission.) The top image shows the photo a camera would take; the middle image shows what someone with full vision would see (as we see clearly only in the center of our vision, what we are looking directly at). The bottom image shows how someone with vision loss from age-related macular degeneration would see it.

To help family members and friends better understand the visual and functional effects of age-related macular degeneration, Macular Degeneration Support has created another online simulation gallery, Through Our Eyes: How People with AMD See.
By Lylas G. Mogk, M.D., Edited by Sefy Paulose, M.D., March, 2022
Risk Factors for AMD
To decrease your risk of developing age-related macular degeneration or to decrease the rate of progression if you already have age-related macular degeneration, here are some actions you can take:
- Don’t smoke – and if you do smoke, try to stop.
- Eat dark green leafy vegetables. These vegetables—such as kale, spinach, and collards—contain lutein, a substance that neutralizes the free radicals that would otherwise damage the macula. Your healthcare provider may recommend a lutein supplement if you take Coumadin and can’t eat these vegetables because of their vitamin K.
- Eat a diet rich in omega-3 fatty acids, which are found in fish, fish oil, flaxseeds, and some nuts. Omega-3 fatty acids reduce inflammation.
- Control your blood pressure and cholesterol levels.
- Exercise regularly and keep your weight down.
The Four Risk Factors We Can’t Control
- Advanced age: Although AMD may occur earlier, studies indicate that people over age 60 are at greater risk than those in younger age groups. This risk increases more than threefold in patients older than 75 compared to the group of patients between 65 and 74 (Beaver Dam Eye Study).
- Caucasian people are much more likely to lose vision from age-related macular degeneration than any other group.
- A gene variant that regulates inflammation: While not all types of macular degeneration are hereditary, specific genes have been strongly associated with a person’s risk of age-related macular degeneration, and genetic predisposition may account for half the cases of age-related macular degeneration in this country.
- Family history: Studies indicate that your chances of developing age-related macular degeneration are three to four times higher if you have a parent, child, or sibling with macular degeneration.
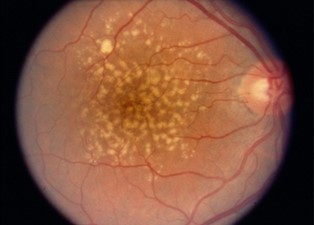
There are two types of AMD: dry (atrophic) and wet (neovascular or exudative). Most AMD starts as the dry type and may progress to the wet type in 10-20% of individuals. Age-related macular degeneration is usually bilateral (i.e., occurs in both eyes) but does not necessarily progress at the same pace in both eyes. It is, therefore, possible to experience the wet type in one eye and the dry type in the other.
What is Dry Macular Degeneration?
The dry (atrophic) type affects approximately 80-90% of individuals with AMD. The dry AMD tends to progress more slowly than the wet type, and there is not yet an approved treatment or cure. In dry age-related macular degeneration, small white or yellowish deposits, called drusen, form on the retina beneath the macula, causing it to deteriorate or degenerate over time.
Drusen is the hallmark of dry AMD. These small yellow deposits beneath the retina are a buildup of waste materials, including cholesterol, protein, and fats. Typically, when drusen first form, they don’t cause any vision loss. However, they are a risk factor for progressing to vision loss.
What is Wet Macular Degeneration?
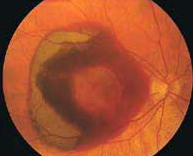
The wet/neovascular type affects approximately 10-15% of individuals with age-related macular degeneration but accounts for approximately 90% of all cases of severe vision loss from the disease. Wet AMD tends to progress rapidly and causes severe loss of central vision. In this form, the degeneration of the macula causes the retina to create a protein called VEGF. This is made by the retina to protect itself and to create new blood vessels. However, this attempt is futile as those new blood vessels are abnormal. In wet AMD, these abnormal blood vessels tend to break, bleed, and leak fluid, which causes damage to the macula. After some time, a scar overlies the entire macula, causing severe loss of central vision.
Barbara Beskind, 91-Year-Old Silicon Valley Tech Designer

Barbara Beskind is 91 years old, a former occupational therapist, has macular degeneration and is a designer with IDEO, a Silicon Valley technology company.
Her advice for the millions of seniors in the United States experiencing age-related vision loss? “The more adaptable, organized, and self-disciplined you’ve been your whole life, the better you will be able to adjust to your needs as they come up. As we get older, we have to expect change and embrace it.”
Learn more about ways to adapt your home following Barbara’s design principles:
- Keep your household organized! Follow the principles of work simplification and energy conservation.
- Use tactile markings and “bump dots” to label various household items.
- Learn to get around safely indoors and outdoors.
- Check out our Getting Started 2020: A Guide for People New to Vision Loss – VisionAware for more ideas to help you live well with low vision.
How is AMD Diagnosed?
As you may not notice any signs of age-related macular degeneration, the best way to protect your sight from AMD is to have regular annual eye examinations. During this examination, an ophthalmologist or optometrist will be able to assess your retina’s health and identify any signs of AMD by doing a comprehensive medical eye examination. The following can help them recognize AMD:
- They will assess your overall health and consider risk factors such as smoking, high blood pressure, and obesity.
- They test your visual acuity to measure the sharpness and clarity of your near and distance vision.
- They will test your vision with different lenses, often using a machine called a phoropter, to determine if regular glasses or contact lenses can improve or correct your vision.
- They will evaluate your visual fields.
- They will examine the anterior segment of your eye, including the cornea, pupil, iris, lens, and aqueous drainage structure, using a special microscope called a slit lamp.
- They will perform a dilated eye exam to evaluate your retina for any signs of AMD.
- They will perform non-invasive imaging called Optical Coherence Tomography (OCT).
Monitoring Vision Changes
Once age-related macular degeneration has been diagnosed, your eye care professional may give you an Amsler Grid to use at home as an early warning system for changes in age-related macular degeneration, particularly a change from dry to wet AMD. This black and white grid looks like graph paper with a small black dot at its center. The macula is particularly sensitive to horizontal and vertical lines; therefore, waviness, distortion, or missing lines on the grid may be noticed before a change in visual acuity.
 Amsler Grid as Seen with Normal Vision
Amsler Grid as Seen with Normal Vision
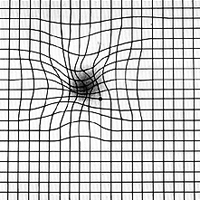 Amsler Grid as Seen with AMD
Amsler Grid as Seen with AMD
Remember: Early detection of vision changes means timely treatment that can save your vision. To find out more, watch this video by the American Academy of Ophthalmology on how to use an Amsler Grid.
What Treatments Are Available for Dry Macular Degeneration?
In dry age-related macular degeneration, small white or yellowish deposits, called drusen, form on the retina beneath the macula, causing it to deteriorate or degenerate over time. There is no mainstay treatment for dry AMD. However, depending on the stage of dry AMD, taking a particular supplement can help slow its progression.
AREDS2
This supplement is called AREDS2. Studies have found that the following formulation can help decrease the progression of dry AMD: 500 milligrams (mg) of vitamin C, 400 international units of vitamin E, 80 mg zinc as zinc oxide, 2 mg copper as cupric oxide (to avoid anemia with high zinc intake), 10 mg lutein and 2 mg zeaxanthin. However, ask your doctor before taking this supplement, as the AREDS2 clinical trial has shown no benefit for mild stages of AMD.
Modifiable Risk Factors
Studies suggest that modifiable risk factors may help slow the progression of AMD or prevent worsening. The most studied and proven modifiable risk factor is smoking. Studies have shown that smokers have an increased risk of dry AMD than nonsmokers. Also, ex-smokers have a decreased risk of dry AMD than current smokers. By stopping smoking, you will help prevent the progression of dry AMD
Certain vegetables contain lutein, which protects the macula from further damage. Thus, studies have shown increasing the quantity of antioxidant-rich foods, such as fresh fruits and dark green leafy vegetables (kale, collard greens, and spinach, for example). Other studies have also suggested eating fatty fish to reduce the severity of dry AMD, as these fish are high in omega-3 fatty acids, which help decrease inflammation.
Obesity and uncontrolled hypertension are two known risk factors that worsen the disease progression of AMD. Because of this, incorporate regular exercise and following with your primary care provider about combating obesity and hypertension.
What Treatments Are Available for Wet Macular Degeneration?
In wet (neovascular/exudative) age-related macular degeneration (AMD), abnormal blood vessels develop under the macula and break, bleed, and leak fluid. This damages the macula and, if left untreated, can result in rapid and severe loss of central vision.
Targeting Vascular Endothelial Growth Factor (VEGF)
The most effective treatments to date for wet AMD are medications that target the protein that causes the abnormal blood vessels to form and vascular endothelial growth factor (VEGF), a protein produced by a damaged retina that causes the production of abnormal blood vessels. However, these blood vessels damage your eye more by causing worsening swelling and bleeding and can cause scars that can pull on your retina and cause a retinal detachment.
Injections
In these cases, your doctor may treat you with an injection of anti-VEGF medicine. Anti-VEGF medicine blocks VEGF, which prevents the growth of new blood vessels in the eye. Although the main role of anti-VEGF injections is to prevent your vision from worsening, this medication can sometimes improve your vision. Your ophthalmologist may prescribe three anti-VEGF medications – Avastin, Lucentis, and Eylea.
Laser
In some very aggressive wet AMD scenarios, your doctor may also recommend laser treatment called photodynamic therapy (cold laser). The purpose of this laser is to slow down progression. Although this laser does not prevent significant vision loss, it may be used in some rare instances of exudative AMD.
Surgical
If there is still a concern for worsening wet AMD, your doctor may suggest surgical treatment. However, these surgical procedures were developed before the invention of anti-VEGF treatments. For the most part, these surgical treatments are rarely used.
By Lylas G. Mogk, M.D., Updated by Sefy Paulose, M.D., March. 2022
Clinical Trials for Macular Degeneration
To receive approval from the U.S. Food and Drug Administration (FDA), a new drug or treatment must be proven safe and effective by undergoing a rigorous series of controlled studies. To ensure accuracy, neither the patient nor the examiners can know which patients received the actual treatment and which were the untreated (or “control”) subjects.
These are called “double-blind” or “double-masked” studies and usually yield the most reliable results. The medication is coded, and patients are randomly placed into the treatment or control group. When the study is concluded, the code is revealed, and it is then possible to determine who received the actual drug and who received the inactive substance or placebo.
As defined by the U.S. National Institutes of Health, most clinical trials are designated as Phase I, II, or III, based on the questions the study is seeking to answer:
- In Phase I clinical trials, researchers test a new drug or treatment in a small group of people (20-80) for the first time to evaluate its safety, determine a safe and effective dosage range, and identify possible side effects.
- In Phase II clinical trials, the study drug or treatment is given to a larger group of people (100-300) to determine its effectiveness and further evaluate its safety.
- In Phase III studies, the study drug or treatment is given to even larger groups of people (1,000-3,000) to confirm its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow the drug or treatment to be used safely.
- In Phase IV studies, after the Food and Drug Administration has approved the drug, continuing studies will determine additional information, such as the drug’s risks, side effects, benefits, and optimal use.
Read More: